Want to enroll participants in a longitudinal or multi-day study? In this tutorial, we’ll show you how in three easy steps.
Step 1: Create Your Studies
The first step is to create one study for each part of the overall experiment. This means that if your experimental design involves collecting baseline data and follow-up data, code separate baseline and follow-up studies on your FindingFive account. If compatible with your design, we recommend that you include a text stimulus in your baseline study, informing participants that they may be invited to complete a follow-up study. This could increase participant retention!
Note that it is important to create separate studies, rather than separate sessions of the same study in order to make full use of FindingFive’s multi-part study tools. If you want your baseline and follow-up studies to contain identical measures, simply make a copy of your baseline study from the “Studies” dashboard, and FindingFive will take care of duplicating all associated stimuli, responses, trial templates, and procedures.
Step 2: Launch Baseline Study
Next, launch a session of your baseline study. When launching, consider selecting the option to access participant contact information (Step 3 of the Session Wizard). This allows you to notify them of their inclusion in your follow-up study — see Notifying Participants section below!
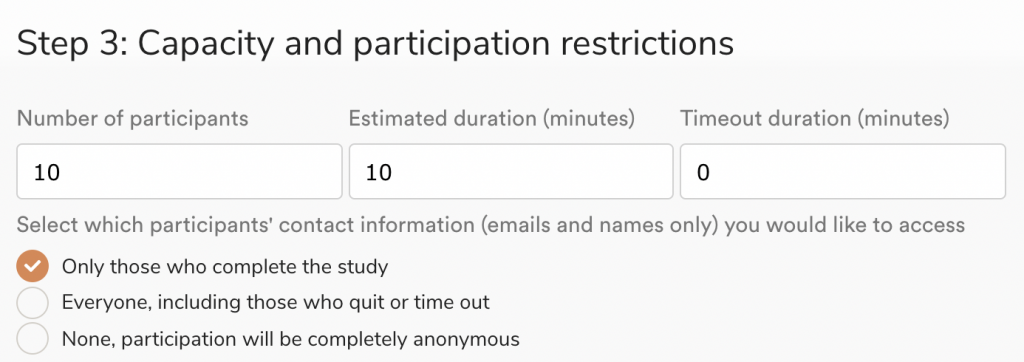
The step of launching your baseline study is straightforward but must occur before you launch your follow-up study. Otherwise, you won’t be able to select the baseline as a prerequisite for the follow-up.
Step 3: Launch Follow-Up Study & Include Prerequisites
You can either launch your follow-up study concurrently with your baseline study, or you can launch it after your baseline session has completed.
Launching concurrently allows participants to complete the follow-up study immediately. For example, you could keep both studies open for a 5-day period; any participant who completes the baseline study in that window will be eligible to complete the follow-up study.
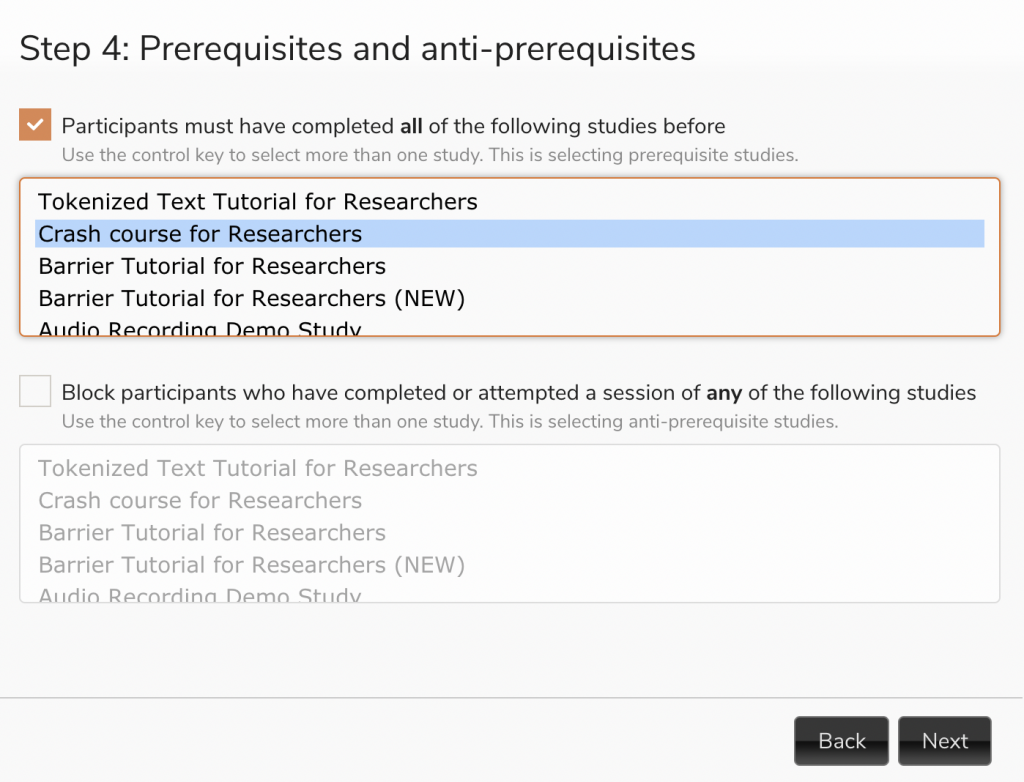
When launching the follow-up, use our Session Wizard to select your baseline study as a prerequisite.
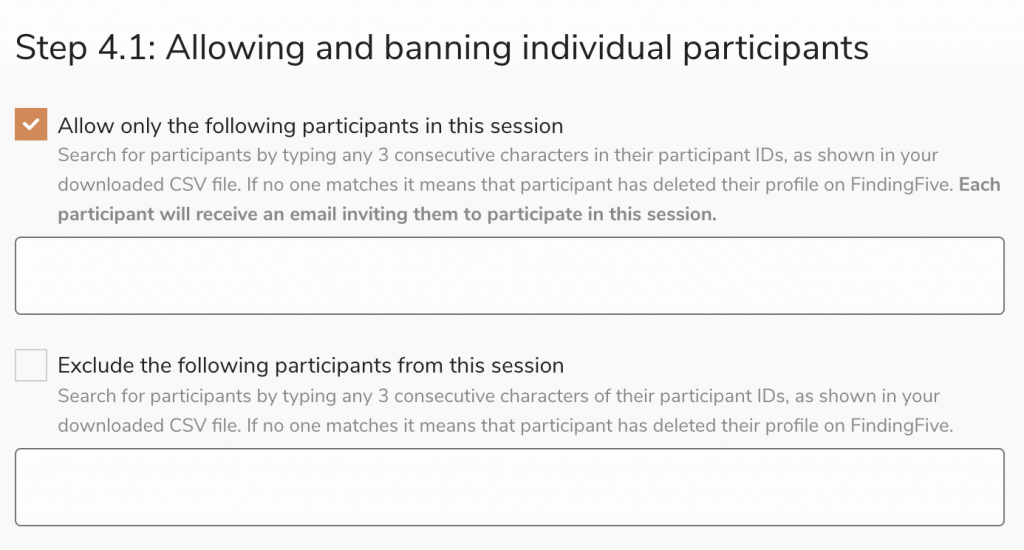
If you instead choose to close the baseline study before launching the follow-up, you can selectively include and exclude participants using Participant ID. Closing the baseline study allows you to download and analyze the data, so you can determine the appropriate participants to include and exclude based on your metrics of choice.
Notifying Participants
For individual participants who are included in a study via Participant IDs (Session Wizard Step 4.1), FindingFive takes care of sending out email notifications inviting them to complete your follow-up study.
We are working on bringing email notifications to inclusion via completion of a previous study (Session Wizard Step 4), and we will update this tutorial when that feature is in place! As a reminder, inclusion via completion of a previous study is only necessary if you want to run baseline and follow-up studies concurrently. For now, we recommend including a text stimulus in your baseline study, alerting participants that they will be eligible for a follow-up study. As an additional measure, you can choose to manually email all participants who’ve completed your baseline study, even while the session is still active (if you chose to access participant contact information when launching your baseline session). Note that though you can access contact information, you cannot access any baseline data while the session is still active, so only use this feature if you want to include all participants from your baseline study in your follow-up study. To manually email participants, please find their information on the baseline study’s “Manage Session” page.
That’s it! We hope you find that launching a multi-part study on FindingFive is simple and easy. As always, please feel free to reach out with questions on our forum or at researcher.help@findingfive.com.
